
eclat

Suitable for arrhythmia testing
Use up 7 days of continuous measurement
Holter electrocardiogram monitor eclat
"24-hour testing is not enough to detect sudden arrhythmias."
I often hear this opinion from the medical field.
"I want to measure for a long time, but the cost is not justified."
"We have a limited number of Holter monitors, so patients have to wait."
To solve these problems, we propose a solution called "disposable."
The electrocardiogram obtained by this device will be examined by the Company cardiologists,
A clinical laboratory technician will analyze the data and prepare an interpretation report, all of which is included in the cost.
It will also be useful in promoting work style reform for medical professionals.

Three features of eclat

●Insurance points Continuous electrocardiogram testing using eclat is covered by insurance.
| D210 | Holter monitoring | 1. For every 30 minutes or fraction thereof | 90 points |
| 2. If it exceeds 8 hours | 1,750 points |
Source: March 5, 2020 Health Insurance Issue No. 0305 No. 4
The value that eclat provides
Patients
Increased sense of security
Improved arrhythmia detection rate provides greater peace of mind.
Reduced burden of visiting hospitals
The test was completed in two visits.
The device can be fitted from the first consultation and can be returned by mail.
Skin-friendly electrode pads
These hypoallergenic electrode pads are gentle on the skin and provide excellent adhesion. They blend easily with the skin, reducing pain and exfoliation when removed.
Bathing is possible during the examination
It is waterproof (equivalent to IPX5) so you can use it in the shower.
QOL is less likely to decrease while wearing the device.
Medical Institutions
Improvement in arrhythmia detection rate
Compared to 24 hours, the detection rate of arrhythmias has improved.
Reducing the burden on medical staff
Our technicians and doctors analyze the ECG data and provide detailed reports to support your medical treatment. There is no need to manage the loan and return of equipment.
Cost reduction and easy cost management
Because it is a disposable product, there are no running costs for maintenance. In addition, the price is set so that it falls within the health point limit even when used as needed.
Reduce lost opportunities
By having it in stock, you won't miss the timing to measure.
3 steps of testing
Step 1.Apply

The device is attached to the examinee's sternum.
Turn on the power button to start the test
*Enter test information on the dedicated website
Staff will attach the sticker to patients in the hospital
Step 2.Mail

After the specified time has passed, the patient
Return the device using a dedicated letter pack
*Letter pack will also be delivered
Patients return it themselves
Step3.Analysis

Analysis by clinical laboratory technicians and cardiologists
View the results on a dedicated website (can be saved as a PDF)
*You will receive a notification email when the analysis is complete.
Kokoromill engineers and doctors analyze
Product Information and Specifications
|
Item |
specification |
|
common name |
Long-term electrocardiogram data recorder (Holter electrocardiogram) |
|
Medical device certification number |
306AFBZX00067000 |
|
Guidance method |
Bipolar induction system 1 channel |
|
Validity period |
Up to 2 years after manufacture |
|
Dimensions and weight etc. |
External dimensions: 45mm(W) x 150mm(D) x 14mm(H) |
|
sampling |
250Hz |
|
Usage/storage environment |
Temperature -10~35°c |

For healthcare professionals
You can log in to eclatWebhere.
gram
gram (discontinued product)
This product is no longer available for sale. Please order the eclat product listed above as its successor.
Three features of gram
Three features of gram
maximum 7 consecutive days do
Possible to obtain electrocardiogram
in one test medical institution
Monetizable pricing model
Management of equipment rental and return
inspection schedule No adjustment required
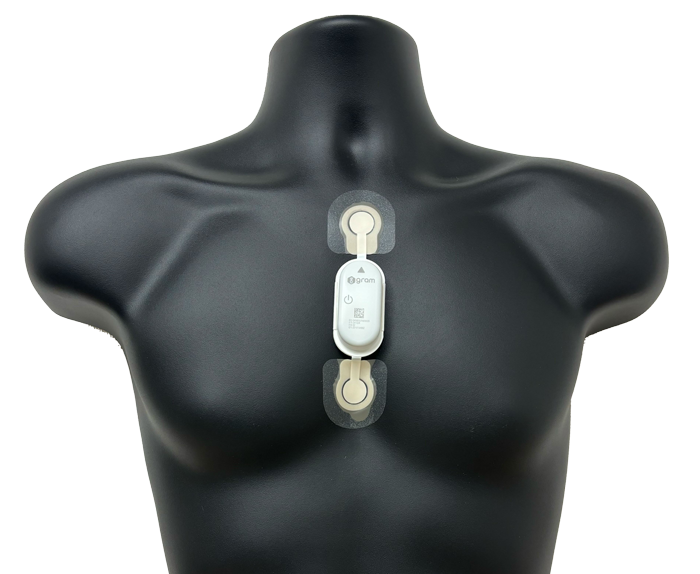
Image of equipment installation
*Non-invasively attached to the chest surface
Main product specifications
|
Item |
specification |
|
common name |
Long-term electrocardiogram data recorder |
| Pharmaceutical approval |
Class 2 controlled medical device |
|
Medical device certification number |
303AFBZX00028000 |
|
External dimensions |
163㎜(W)×33㎜(D)×14.5㎜(H) |
|
Interelectrode distance |
114㎜ |
|
weight |
Approximately 19g |
|
Manufacturer/seller |
durantis Co., Ltd. |

Inspection flow ~3 easy steps~


1Attach the device to the patient
The device is pasted on the sternum of the test taker,
Turn on the power button to start the test.
*Enter test information on the dedicated website


2Returnthe device by mail
After the specified time has elapsed, the test taker will return the
Put the equipment in and drop it in the mailbox.
*Return to Kokoro Mill's analysis center

3Analyzethe data
The data of the collected equipment will be sent to Kokoro Mill Co., Ltd.
Analyzed by clinical laboratory technicians and affiliated physicians.
*You will receive a notification email when the analysis is finished.
*Analysis results can be viewed on the dedicated website and saved as PDF.
Overview of Kokoro Mill
Kokoro Mill Co., Ltd. is a company that operates electrocardiogram analysis services using wearable electrocardiograms. The company operates and develops electrocardiogram measurement services using the Holter electrocardiograph ``gram'' for medical institutions and electrocardiogram analysis services using the wearable electrocardiograph ``duranta'' for companies and individuals.
Specialist doctors and clinical laboratory technicians analyze the evaluation report of the acquired electrocardiogram data and provide a report on arrhythmia, stress, sleep quality, and sleep apnea syndrome.
| Established | November 2021 |
| Head Office | 2F Kanno Building, 6-2-16 Nishi-Shinjuku, Shinjuku-ku, Tokyo |
| Board Director | Daiki Hayashi |
| website | https://kokoromil.com/ |
Business
- Health information service that analyzes autonomic nerves and arrhythmia
- Medical equipment rental business
- Information collection, processing and management of electrocardiogram data
- Electrocardiogram data analysis work
- Healthcare related business
Inquiry
In charge of Macnica Kokoro Mill
- E-mail:kokoromil@macnica.co.jp
